![Stereoselective Synthesis of 12-Tetrazolyl Substituted (E)-5H-Quinazolino[3,2-a]quinazolines via Sequential Ugi-Azide/Staudinger/aza-Wittig/Addition/Ag(I)-Catalyzed Cyclization | The Journal of Organic Chemistry Stereoselective Synthesis of 12-Tetrazolyl Substituted (E)-5H-Quinazolino[3,2-a]quinazolines via Sequential Ugi-Azide/Staudinger/aza-Wittig/Addition/Ag(I)-Catalyzed Cyclization | The Journal of Organic Chemistry](https://pubs.acs.org/cms/10.1021/acs.joc.2c02621/asset/images/large/jo2c02621_0001.jpeg)
Stereoselective Synthesis of 12-Tetrazolyl Substituted (E)-5H-Quinazolino[3,2-a]quinazolines via Sequential Ugi-Azide/Staudinger/aza-Wittig/Addition/Ag(I)-Catalyzed Cyclization | The Journal of Organic Chemistry

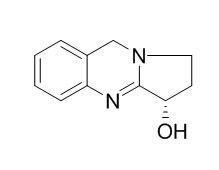
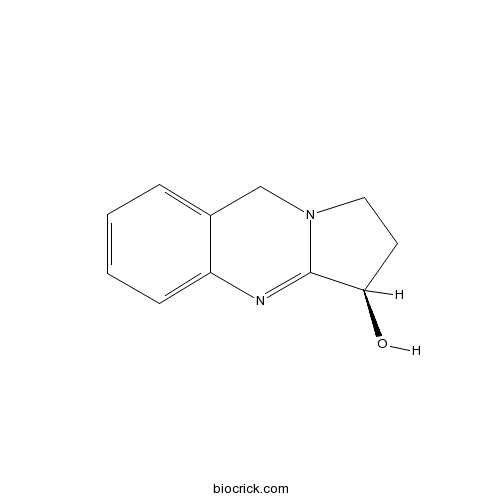
Exploring Derivatives of Quinazoline Alkaloid l-Vasicine as Cap Groups in the Design and Biological Mechanistic Evaluation of Novel Antitumor Histone Deacetylase Inhibitors | Journal of Medicinal Chemistry

Exploring Derivatives of Quinazoline Alkaloid l-Vasicine as Cap Groups in the Design and Biological Mechanistic Evaluation of Novel Antitumor Histone Deacetylase Inhibitors | Journal of Medicinal Chemistry

Exploring Derivatives of Quinazoline Alkaloid l-Vasicine as Cap Groups in the Design and Biological Mechanistic Evaluation of Novel Antitumor Histone Deacetylase Inhibitors | Journal of Medicinal Chemistry
![PDF] Synthesis of Ofornine mimics from natural product l-vasicine as anti-hypertensive agents. | Semantic Scholar PDF] Synthesis of Ofornine mimics from natural product l-vasicine as anti-hypertensive agents. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/805e3c20bd9bac728a86c88e4e034caf1b109882/3-Table1-1.png)
PDF] Synthesis of Ofornine mimics from natural product l-vasicine as anti-hypertensive agents. | Semantic Scholar
Liquid Chromatography Coupled to Mass Spectrometry Based Identification of Elite Chemotypes of Adhatoda vasica Nees for Profitab

PDF) Development and validation of an HPLC-DAD method for Rapid quantification of vasicine in Adhatoda vasica leaves and commercial products
Development and validation of an HPLC-DAD method for Rapid quantification of vasicine in Adhatoda vasica leaves and commercial p

PDF) Metal-free transfer hydrogenation of nitroarenes in water with vasicine: revelation of organocatalytic facet of an abundant alkaloid | manoranjan kumar and Sushila Sharma - Academia.edu

Exploring Derivatives of Quinazoline Alkaloid l-Vasicine as Cap Groups in the Design and Biological Mechanistic Evaluation of Novel Antitumor Histone Deacetylase Inhibitors | Journal of Medicinal Chemistry

Exploring Derivatives of Quinazoline Alkaloid l-Vasicine as Cap Groups in the Design and Biological Mechanistic Evaluation of Novel Antitumor Histone Deacetylase Inhibitors | Journal of Medicinal Chemistry

PDF) Development and validation of an HPLC-DAD method for Rapid quantification of vasicine in Adhatoda vasica leaves and commercial products
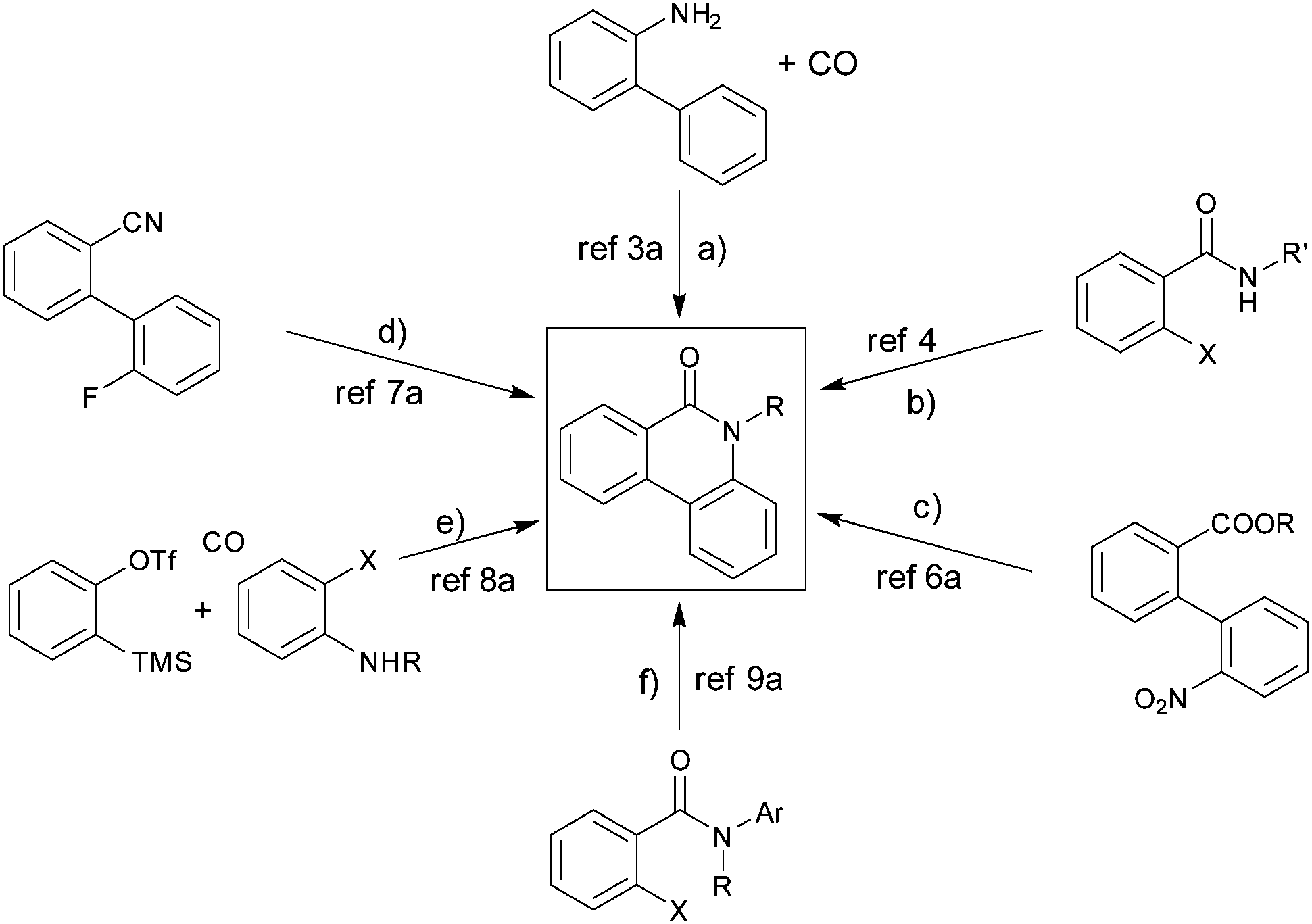
Microwave assisted synthesis of phenanthridinones and dihydrophenanthridines by vasicine/KO t Bu promoted intramolecular C–H arylation - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C6OB01362G

PDF) Exploring derivatives of quinazoline alkaloid l-vasicine as cap groups in the design and biological mechanistic evaluation of novel antitumor histone deacetylase inhibitors | Abdul Rouf - Academia.edu
![PDF] Synthesis of Ofornine mimics from natural product l-vasicine as anti-hypertensive agents. | Semantic Scholar PDF] Synthesis of Ofornine mimics from natural product l-vasicine as anti-hypertensive agents. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/805e3c20bd9bac728a86c88e4e034caf1b109882/6-Figure2-1.png)
PDF] Synthesis of Ofornine mimics from natural product l-vasicine as anti-hypertensive agents. | Semantic Scholar
Quercetin Dihydrate | CAS:6151-25-3 | PLA2 and PI 3-kinase inhibitor | Flavonoids | High Purity | Manufacturer BioCrick